David Heymann is Professor of Infectious Disease Epidemiology at the London School of Hygiene & Tropical Medicine and Head of the Centre on Global Health Security (CGHS) at Chatham House. In this post, compiled from a Chatham House CGHS report drafted by Gemma Buckland, he describes the current status of antibiotic stewardship in animals. The Chatham House report on Tackling Antibiotic Resistance for Greater Global Health Security can be accessed here.
Bacteria (including those that are resistant) can be spread from animals to humans by handling and eating raw or inadequately cooked food, or through direct animal contact. In addition, bacteria from animals can be spread to humans through manure that contaminates water, soil, and plants.
Antibiotics are used in animal husbandry to prevent infection in animal stocks (prophylaxis), to prevent the spread of an infection in a small group of animals to others in the stock (metaphylaxis), and as additives in feed to promote faster growth. Some antibiotics that are used in food animals are also important in human medicine, and these uses of antibiotics in animals are all are thought to provide favourable conditions for resistance. Evidence from the last 35 years suggests a correlation between antibiotic use in animal husbandry, mainly livestock, poultry, and aquaculture, and the rise and spread of antibiotic resistance genes in human pathogens, as well as direct transfer of resistant bacteria from animals to humans.

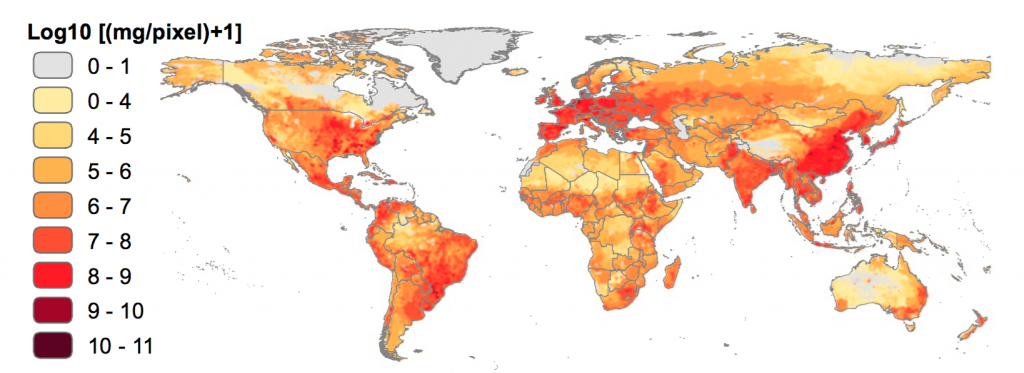
Approximately 50% of all antibiotics used worldwide are used in raising animals, including poultry, and fish. In some European countries, even where antibiotic use is restricted to the treatment of known infections in animals and fish, up to 180 mg of antibiotics are used to produce one kilogram of meat. In other countries, including the United States, antibiotics are used as additives to animal and feed to prevent infection and enhance growth, sometimes as a replacement for agriculture practices that expose animals to increased risk of infection. Antibiotics for animal husbandry are often used with little or no consultation with veterinarians, because many antibiotics used for veterinary purposes are available over-the-counter and not subjected to medicines regulation. Disease diagnosis and treatment are therefore often done by farmers themselves, and may result in higher levels of antibiotic misuse.
The World Organisation for Animal Health (OIE), the Food and Agriculture Organization of the United Nations (FAO) and the World Health Organization (WHO) have begun to jointly examine the interface between animals and humans, and its implications for antibiotic resistance. A number of strategies have been recommended to help tackle resistance. These include:
- Supporting global implementation and harmonization of standards;
- Improving surveillance and monitoring of antibiotic use;
- Advocating for the International Cooperation on Harmonization of Technical Requirements for Registration of Veterinary Medicinal Products guidelines to ensure the quality of veterinary medicinal products; and
- Supporting lower and middle income countries to strengthen their veterinary services.
Pharmaceutical companies and veterinarians often profit from antimicrobial sale and use; however, there are no conclusive data to demonstrate that this affects the prescribing practices of veterinarians. In countries with regulatory systems for veterinary medicines, enforcement and adherence varies. Denmark placed restrictions on the degree to which veterinarians can profit from prescriptions. Several national veterinary organizations, such as the American Veterinary Medical Association, have developed prudent-use guidelines based on regulations. Mandatory restrictions and prudent-use guidelines could benefit both human and animal health if widely adopted and enforced, but their effectiveness has not yet been evaluated.
Regulation in animal husbandry and the agriculture sectors is complex, because of the cross-cutting nature of the authorities and industries involved. This complexity can be a barrier to effective antibiotic policy formation and implementation. The concept of ‘One Health’, to expand interdisciplinary collaboration and communication in health care for humans, animals and the environment, is thought to provide the best opportunity to tackle these barriers.
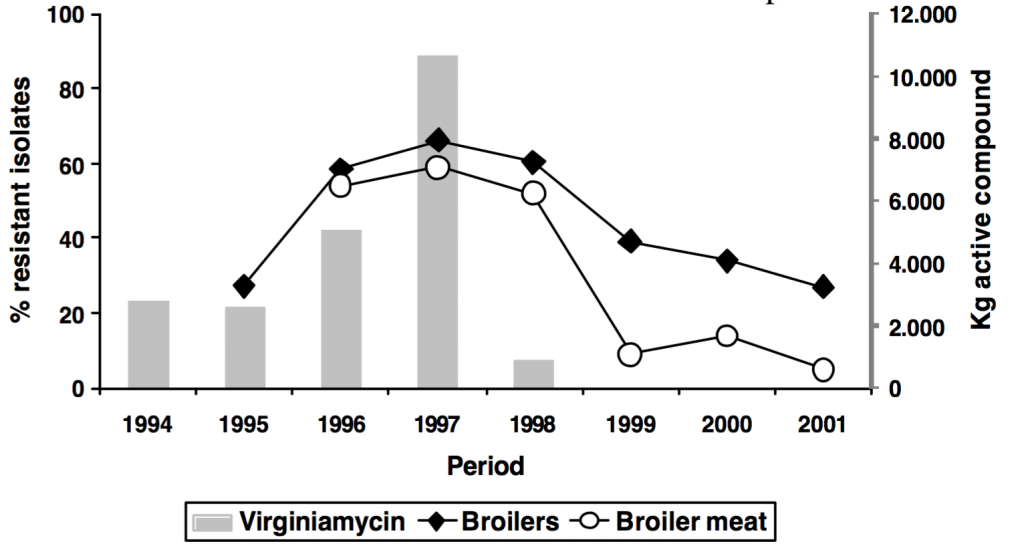
European countries have banned the use of antibiotics as growth promoters in agriculture since the late 20th century. In Denmark, this decrease in non-specific antibiotic use has had no adverse effect on animal mortality, although the economic effects of the ban in Denmark and other European countries have not yet been fully documented. In New Zealand, all antibiotics for use in animals must be registered and approved by the New Zealand Safety Authority, and they cannot be used unless there is a veterinary prescription. Only approved traders are allowed to sell drugs, and they may not be promoted or advertised to the public. Some recommend restricting the use, in food production, of any antibiotic of critical importance to human health. In the United States, legislation has been introduced to eliminate the non-specific use of such critical antibiotics in animals, including as growth promoters, but the addition of other antibiotics to animal feed continues. Policy options for limiting the use of these critical antibiotics, in addition to legislation, include high user fees and targeted bans. WHO has recently produced a list of antibiotics that are critical for human use, as guidance to the veterinary community and policy makers.
Members of agricultural and allied industries have raised concerns that restrictions on the use of antimicrobials in food and animal production will decrease incentives for new veterinary antibiotic production. They also raise concerns about decreased efficiency of food production because of an increase in infections in animals, and the need for prophylaxis of other animals with which they have contact. Others argue, however, that restrictions could result in no change to animal health or production efficiency. An important leverage point in working with the animal and agriculture sector is engaging the food industry on more prudent antibiotic use, but this has not yet been successful, and in fact has become highly politicised.
Politicisation has played out in inter-country requirements regarding acceptable drug use and availability of drugs to food producers. The EU banned the use of antimicrobial rinses of poultry, which effectively shuts out US poultry imports. Russia has refused food imports from the EU due to trace amounts of certain antibiotics being present. In order to be effective and have real impact, such restrictions need to be global in nature and underpinned by a strong evidence base.

WHO-recommended principles for the containment of antimicrobial resistance in animals intended for food include introducing pre-licensing safety evaluation of antibiotics used in animals that require consideration of potential resistance in humans. Infection prevention and control also has a role in animal husbandry. Alternatives to the use of antibiotics include improved management practices with better hygiene; identifying and mitigating local risk factors for infection through such interventions as vaccination; and potentially, the introduction of probiotics into feed. However, research in this area is underfunded and neglected, and policies and management tools to facilitate the prudent use of antibiotics in the animal industry and animal waste management are underdeveloped, partly because of the lack of a robust evidence base, and partly because of a paucity of studies to demonstrate the cost-effectiveness of interventions.
