Dr. Nicola J Williams is professor of Zoonotic Bacterial Disease at the Department of Epidemiology and Population Health, Institute of Infection and Global Health, University of Liverpool, UK. Her research interests include antibiotic resistance, transmission of food-borne pathogens, and the molecular epidemiology of methicillin-resistant Staphylococcus bacteria in wildlife, livestock and companion animals.
MRSA, or methicillin-resistant Staphylococcus aureus is a major cause of clinical infections in people, usually in hospitals, but also more rarely in the community. The main difficulties when treating such infections is that MRSA is usually multi-drug resistant, that means it is resistant to treatment by multiple classes of antibiotic, but it is not resistant to all antibiotics.
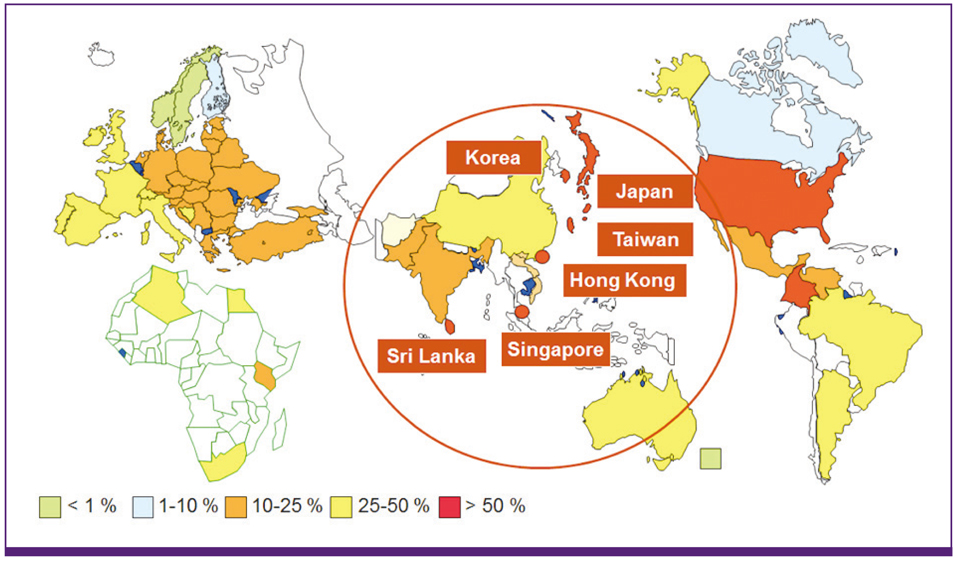
In 2004, a new variant of MRSA was identified in the Netherlands which was traced back to people working on pig farms and which identified livestock as a source, livestock-associated MRSA (LA-MRSA). Since then, there have been numerous reports across Europe and around the world of LA-MRSA being found in pigs, cattle and poultry, and their food products with reports of it also causing infections in people.

For the general public the risk of LA-MRSA is likely to be low, the amount of bacteria on meat is likely to be at a very low level, in other words comprising of very few bacteria on the meat in order to infect people. This is in contrast to foodborne pathogens such as Campylobacter, which cause a significant burden of gastrointestinal disease in people in the developed and developing world, however such bacteria are found in the gastrointestinal tract of animals and colonise to very high levels. Therefore, there is much more opportunity for these types of bacteria to contaminate the surface of the meat during slaughter and processing, and subsequently to infect people. Studies in European countries with a known high prevalence of LA-MRSA in livestock suggest that this type of MRSA is responsible for a small burden of disease in people. However, we must acknowledge that there may be differences in the epidemiology and disease burden caused by these bacteria in developing countries, where there may be different cultures regarding the production of animals, the processing and consumption of meat. Nevertheless, good hygiene in the kitchen when preparing and handling raw meat, washing hands thoroughly and cooking meat properly should remove the risk from both MRSA and also of foodborne pathogens.
In addition, if LA-MRSA, or indeed any other type of MRSA is transmitted via food or animal contact, it does not mean that it will necessarily cause a clinical infection, it can be carried in the nasal passages without causing harm. However, those colonised with MRSA are more likely to get a subsequent infection if for example they go into hospital for an operation, than those who are not colonised.
The greatest risk for colonisation will be those individuals who have direct contact with livestock, so farmers, vets and those working in slaughterhouses. Hygiene is also important among such workers, so those in contact with animals or their products should thoroughly wash their hands and ensure all cuts and wounds are covered to prevent bacteria accessing them. Individuals who have close contact with livestock should inform healthcare providers, if they are to undergo elective surgery or if they are having ongoing treatment which may make them more susceptible to infection. It is already established from a number of studies that veterinarians and farmers are more likely to be carriers of MRSA than the general public, so represent a at risk group.
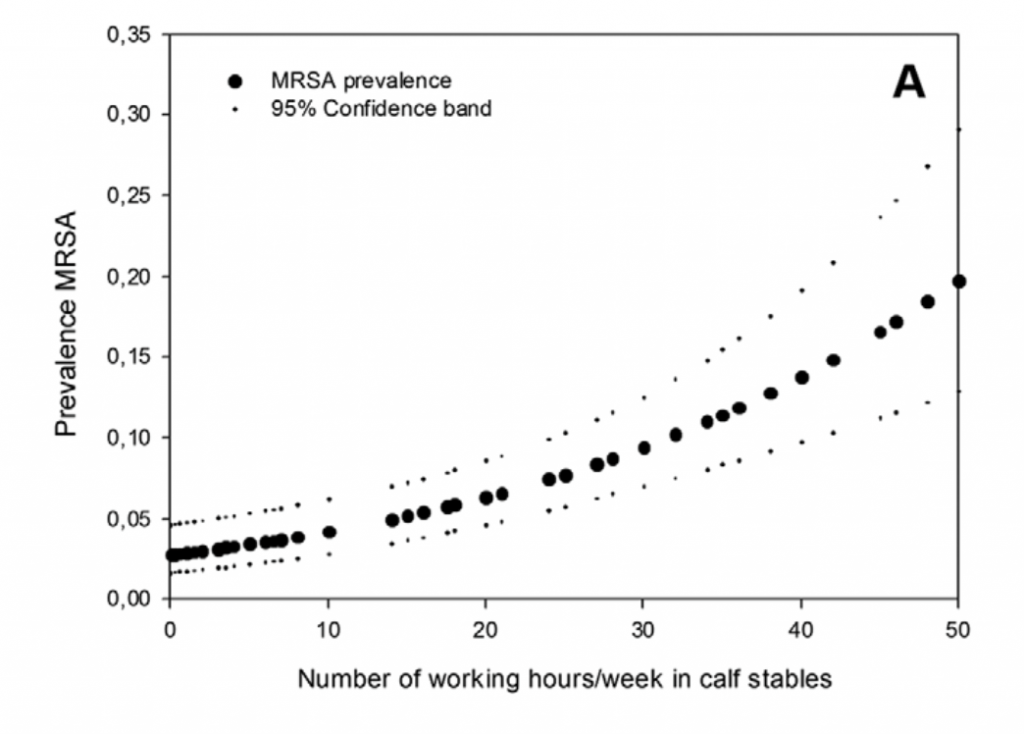
Continued surveillance of LA-MRSA in livestock is important. Equally, surveillance of antibiotic use is also crucial in livestock systems and should be used to inform intervention strategies to limit antibiotic use and to also monitor their success. Reducing the unnecessary use of antibiotics is vital in reducing the selective pressure in livestock for such resistant bacteria. Global initiatives around use in agriculture are lacking, and perhaps we need targets for how much we should be using in livestock production, which countries can agree on, similar to what has been devised for tackling climate change. Such targets would have to take into account local variation in farming methods, regulation of antibiotic sales and the ability to monitor use. However, if we are to truly tackle the issue of antibiotic resistance, concerted action is also required around reducing antibiotic use in human medicine.