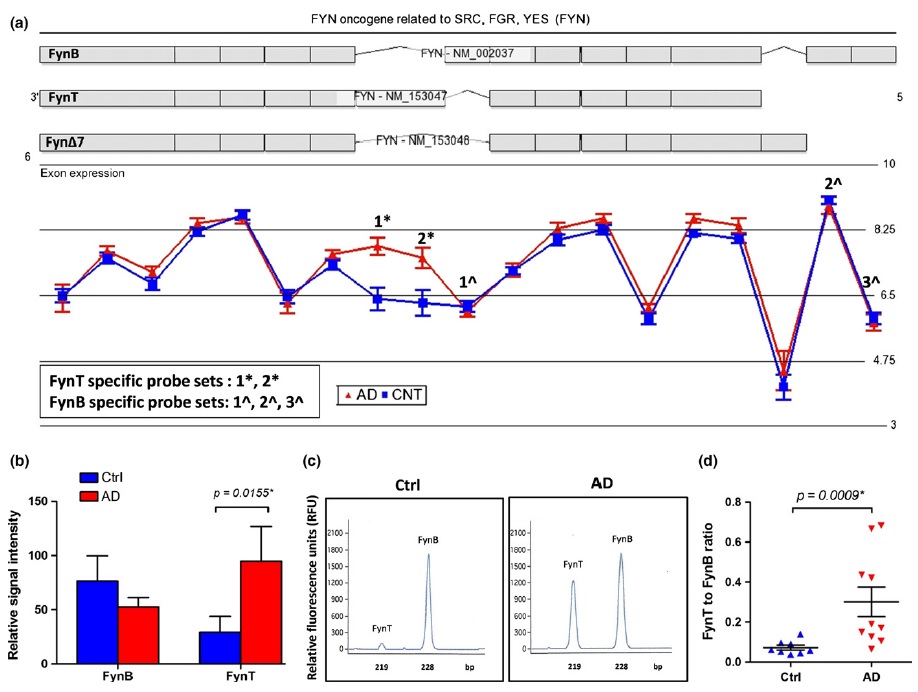
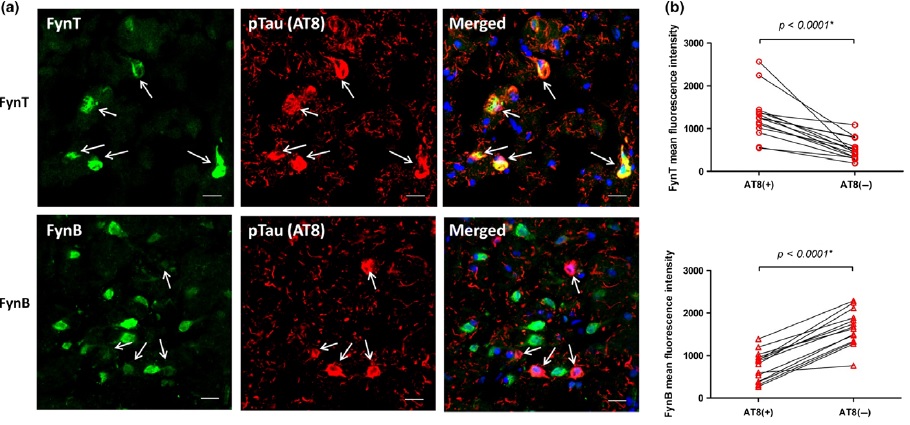
Hot Off the Press: Growth differentiation factor-15 and white matter hyperintensities in cognitive impairment and dementia
Vascular pathology plays an important role in the development of cognitive decline and dementia. In this context, growth differentiation factor-15 (GDF-15) has been suggested to be a biomarker due to its regulatory roles in inflammatory and trophic responses during tissue injury. However, limited data exist on the associations of GDF-15 with either cerebrovascular disease (CeVD) burden or the spectrum of cognitive impairment. Therefore, we aimed to study peripheral levels of GDF-15 incognitive impairment no dementia (CIND) or Alzheimer disease (AD) subjects assessed for CeVD using a case–control cohort design, with cases recruited from memory clinics and controls from memory clinics and the community. All subjects underwent detailed neuropsychological assessment, 3-Tesla magnetic resonance imaging, and venous blood draw. Subjects were classified as CIND or AD based on clinical criteria, while significant CeVD was defined as the presence of cortical infarcts and/or 2 lacunes or more, and/or confluent white matter hyperintensities (WMHs) in 2 or more brain regions. A total of 324 subjects were included in the study, of whom 80 had no cognitive impairment, 144 CIND and 100with AD. Higher GDF-15 levels were significantly associated with disease groups, especially in the presence of CeVD, namely, CIND with CeVD (odds ratios [OR]: 7.21; 95% confidence interval [CI]: 2.14–24.27) and AD with CeVD (OR: 21.87; 95% CI: 2.01–237.43). Among the different CeVD markers, only WMH was associated with higher GDF-15 levels (OR: 3.97; 95% CI: 1.79–8.83). The associations between GDF-15 and cognitive impairment as well as with WMH remained significant after excluding subjects with cardiovascular diseases. In conclusion, we showed that increased GDF-15 may be a biomarker for CIND and AD in subjects with WMH.
Reference
Chai YL, Hilal S, Chong JP, Ng YX, Liew OW, Xu X, Ikram MK, Venketasubramanian N, Richards AM, Lai MK, Chen CP (This paper).


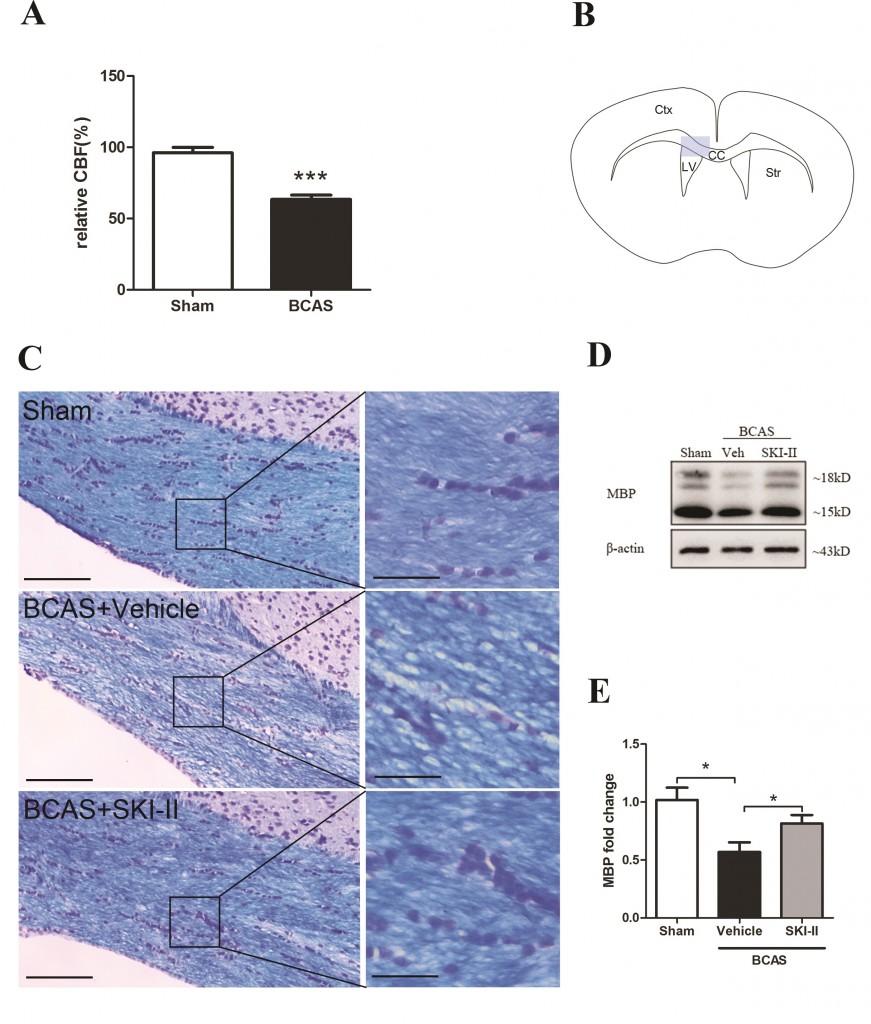
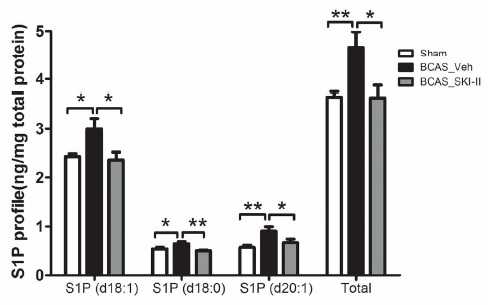 Our results indicated that BCAS induced hypoxia inducible factor (HIF)-1α, Sphk2, S1P, and NG2 up-regulation together with accumulation of WML. In contrast, BCAS mice treated with the SphK inhibitor, SKI-II, showed partial reversal of SphK2, S1P and NG2 elevation and amelioration of WML. In an in vitro model of hypoxia, SKI-II reversed the suppression of OPC differentiation. Our study suggests a mechanism for hypoperfusion-associated WML involving HIF-1α-SphK2-S1P-mediated disruption of OPC differentiation, and proposes the SphK signaling pathway as a potential therapeutic target for white matter disease.
Our results indicated that BCAS induced hypoxia inducible factor (HIF)-1α, Sphk2, S1P, and NG2 up-regulation together with accumulation of WML. In contrast, BCAS mice treated with the SphK inhibitor, SKI-II, showed partial reversal of SphK2, S1P and NG2 elevation and amelioration of WML. In an in vitro model of hypoxia, SKI-II reversed the suppression of OPC differentiation. Our study suggests a mechanism for hypoperfusion-associated WML involving HIF-1α-SphK2-S1P-mediated disruption of OPC differentiation, and proposes the SphK signaling pathway as a potential therapeutic target for white matter disease.
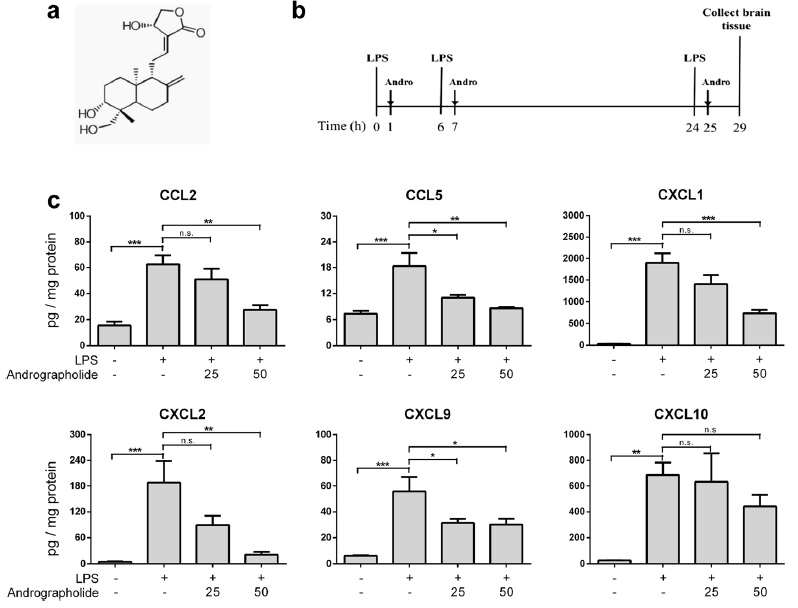
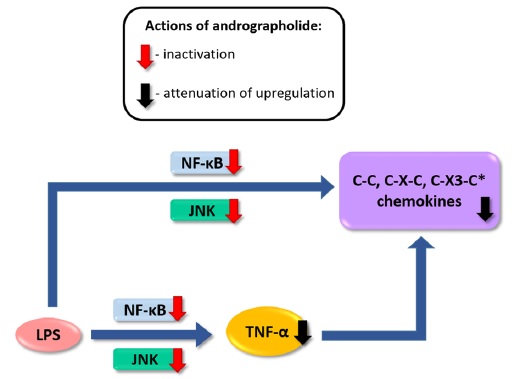 induced chemokine up-regulation both in mouse cortex and in cultured primary astrocytes were measured, including cytokine profiling, gene expression, and, in cultured astrocytes, activation of putative signaling regulators. We found that orally administered andrographolide significantly attenuated mouse cortical chemokine levels from the C-C and C-X-C subfamilies. Similarly, andrographolide abrogated a range of LPS-induced chemokines as well as tumor necrosis factor (TNF)-α in astrocytes. In astrocytes, the inhibitory actions of andrographolide on chemokine and TNF-α up-regulation appeared to be mediated by nuclear factor-κB (NF-κB) or c-Jun N-terminal kinase (JNK) activation. These results suggest that andrographolide may be useful as a therapeutic for neuroinflammatory diseases, especially those characterized by chemokine dysregulation.
induced chemokine up-regulation both in mouse cortex and in cultured primary astrocytes were measured, including cytokine profiling, gene expression, and, in cultured astrocytes, activation of putative signaling regulators. We found that orally administered andrographolide significantly attenuated mouse cortical chemokine levels from the C-C and C-X-C subfamilies. Similarly, andrographolide abrogated a range of LPS-induced chemokines as well as tumor necrosis factor (TNF)-α in astrocytes. In astrocytes, the inhibitory actions of andrographolide on chemokine and TNF-α up-regulation appeared to be mediated by nuclear factor-κB (NF-κB) or c-Jun N-terminal kinase (JNK) activation. These results suggest that andrographolide may be useful as a therapeutic for neuroinflammatory diseases, especially those characterized by chemokine dysregulation.