Hot Off the Press: Andrographolide induces Nrf2 and heme oxygenase 1 in astrocytes by activating p38 MAPK and ERK
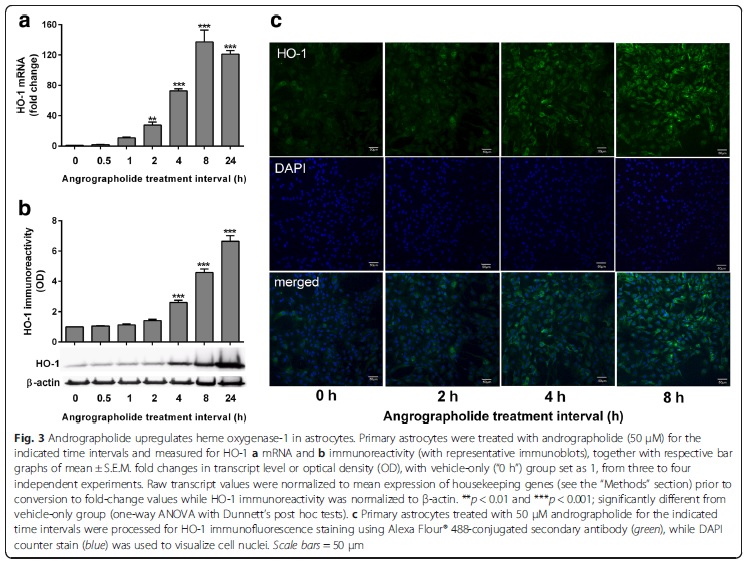 Andrographolide is the major labdane diterpenoid originally isolated from Andrographis paniculata and has been shown to have anti-inflammatory and antioxidative effects. However, there is a dearth of studies on the potential therapeutic utility of andrographolide in neuroinflammatory conditions. Continuing our previous work showing the anti-chemokine effects of andrographolide (Wong et al. 2014; 2016), we investigated the mechanisms underlying andrographolide’s effect on the expression of anti-inflammatory and antioxidant heme oxygenase-1 (HO-1) in primary astrocytes. Measurements of the effects of andrograholide on antioxidant HO-1 and its transcription factor, Nrf2, include gene expression, protein turnover, and activation of putative signaling regulators, p38 and ERK, were carried out. We found that andrographolide potently activated Nrf2 and also upregulated HO-1 expression in primary astrocytes. Interestingly, andrographolide’s effects on Nrf2 seemed to be biphasic, with acute (within 1 h) reductions in Nrf2 ubiquitination efficiency and turnover rate, followed by upregulation of Nrf2 mRNA between 8 and 24 h. The acute regulation of Nrf2 by andrographolide seemed to be independent of Keap1 and partly mediated by p38 MAPK and ERK signaling. These data provide further insights into the mechanisms underlying andrographolide’s effects on astrocyte-mediated antioxidant, and anti-inflammatory responses and support the further assessment of andrographolide as a potential therapeutic for neurological conditions in which oxidative stress and neuroinflammation are implicated.
Andrographolide is the major labdane diterpenoid originally isolated from Andrographis paniculata and has been shown to have anti-inflammatory and antioxidative effects. However, there is a dearth of studies on the potential therapeutic utility of andrographolide in neuroinflammatory conditions. Continuing our previous work showing the anti-chemokine effects of andrographolide (Wong et al. 2014; 2016), we investigated the mechanisms underlying andrographolide’s effect on the expression of anti-inflammatory and antioxidant heme oxygenase-1 (HO-1) in primary astrocytes. Measurements of the effects of andrograholide on antioxidant HO-1 and its transcription factor, Nrf2, include gene expression, protein turnover, and activation of putative signaling regulators, p38 and ERK, were carried out. We found that andrographolide potently activated Nrf2 and also upregulated HO-1 expression in primary astrocytes. Interestingly, andrographolide’s effects on Nrf2 seemed to be biphasic, with acute (within 1 h) reductions in Nrf2 ubiquitination efficiency and turnover rate, followed by upregulation of Nrf2 mRNA between 8 and 24 h. The acute regulation of Nrf2 by andrographolide seemed to be independent of Keap1 and partly mediated by p38 MAPK and ERK signaling. These data provide further insights into the mechanisms underlying andrographolide’s effects on astrocyte-mediated antioxidant, and anti-inflammatory responses and support the further assessment of andrographolide as a potential therapeutic for neurological conditions in which oxidative stress and neuroinflammation are implicated.
References
Wong SY, Tan MG, Wong PT, Herr DR, Lai MK (This paper)
Wong SY, Chan SJ, Wong WS, Wong PT, Lai MK (2014) Andrographolide attenuates interleukin-1β-stimulated upregulation of chemokine CCL5 and glial fibrillary acidic protein in astrocytes. Neuroreport 25:881-6.
Wong SY, Tan MG, Banks WA, Wong WS, Wong PT, Lai MK (2016) Andrographolide attenuates LPS-stimulated up-regulation of C-C and C-X-C motif chemokines in rodent cortex and primary astrocytes. J Neuroinflammation 13:34.
